User Requirements
User requirements are just what the name implies. They are requirements set by the end user. These requirements express how a facility, equipment or process should perform in terms of the product to be manufactured, required throughput, and conditions in which product should be made. User requirements provide information that serves as the basis for further specification, design, and verification of a manufacturing system (i.e., the design solution from the vendor to meet the user requirements which is evaluated during the design review/qualification process). Commissioning and Qualification activities should be structured such that at the end of the Commissioning and Qualification process there is documented evidence that demonstrates that the user requirements have been met.
Note: A common question fielded by PV team members from our customers is how much detail should be in the User Requirement? Some companies write very detailed user requirements often including not only what is needed but why it is needed and how the requirement is to be met. Other companies provide only a high level requirement allowing the vendors to provide a design solution which is then refined through an interrogative design review process. While either option is acceptable, if possible it may be best to allow the equipment manufacturers to provide the design solution.
ISPE separates user requirements into two categories: Process versus General Requirements. Process User Requirements (PURs) are related to product quality and/or GMP (Good Manufacturing Practices) regulatory compliance. These requirements must be qualified, or verified as present and operating per design, and documented in documentation that is approved by the Quality Unit.
General User Requirements are not related to product quality or GMP. These requirements may be critical to personnel safety and/or non-GMP regulatory compliance and should be verified during commissioning activities.
Examples of Process and General User Requirements are provided in Table (ISPE, 2011).
Table 1 – Process User Requirements versus General User Requirements
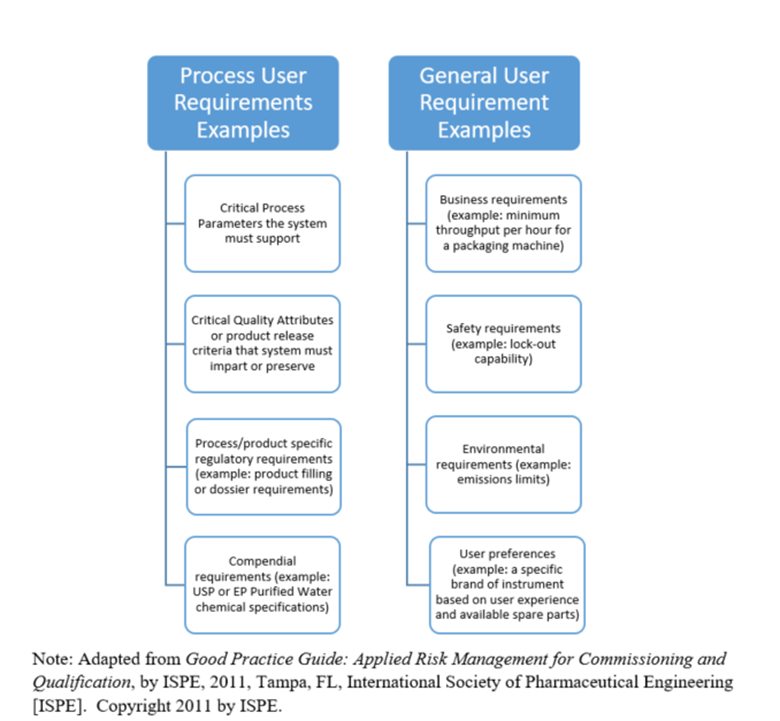
Process User requirements should include the Critical Quality Attributes (CQA) for the product and Critical Process Parameters supported by the system. A CQA is defined by ICH Q8 (2009, p. 11) “… a physical, chemical, biological, or microbiological property or characteristic that should be within an appropriate limit, range or distribution to ensure the desired product quality.” CQAs are typically defined by entities outside of the C&Q team and should be based upon product and process knowledge and understanding, regulatory requirements, and company quality requirements. The product and process knowledge utilized for CQA identification should be based on scientific data.
ICH Q8 (2009, p. 16) defines a Critical Process Parameter as “A process parameter whose variability has an impact on a critical quality attribute and therefore should be monitored or controlled to ensure the process produces the desired quality. A CPP requires the preexistence of a CQA. Examples of a critical process parameter could be temperature of a water system. This parameter is set such that the critical quality attribute, microbial count, is within an acceptable range.
Critical aspects of manufacturing systems are defined in ASTM International (2013) as functions, features, abilities, and performance or characteristics necessary for the manufacturing process and systems to ensure consistent product quality and patient safety. They can be determined through the use of risk assessment tools. ASTM recommends these aspects should be verified. Utilizing an ISPE approach, these aspects will be qualified.
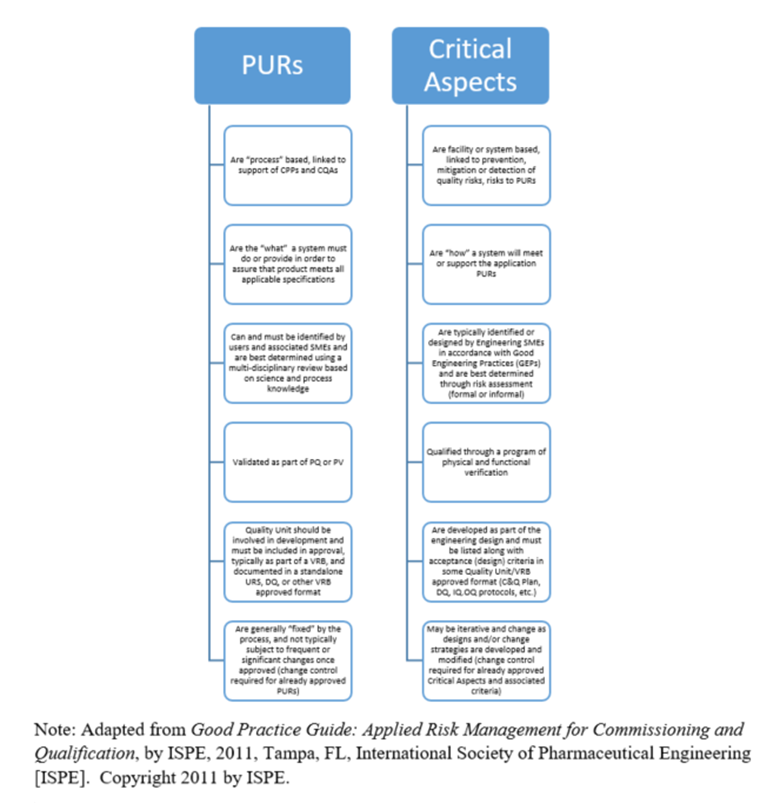
When creating user requirements, it is useful to understand the difference between Process User Requirements and Critical aspects. Examples of these differences are provided in Table 2 (ISPE, 2011).
Table 2 – PURs vs. Critical Aspects
As with many aspects of Commissioning and Qualification, there is no absolute right and wrong in creating User Requirements. The guidelines above help to reduce the time, energy, and costs associated with C&Q activities. User requirements can be as broad or as specific as the client deems necessary. No matter how general or specific, Commissioning and Qualification activities should result in assuring that the requirements have been adequately met.
Have a question on User Requirements or need help? Please use our Contact Us form.
References:
ASTM International. (2013). E2500-13: Standard guide for specification, design, and verification of pharmaceutical and biopharmaceutical manufacturing systems and equipment. West Conshohocken, PA: ASTM International.
International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use [ICH]. (2009). Pharmaceutial development: Q8. Retrieved from https://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q8_R1/Step4/Q8_R2_Guideline.pdf
ISPE. (2011). Good practice guide: Applied risk management for commissioning and qualification. Tampa, FL: International Society of Pharmaceutial Engineering.